Can you smell it too? The odour of asparagus in urine, and the receptor you didn’t know you had.
Every spring, alongside the first seasonal vegetables, asparagus makes its return. Diuretic and fibre-rich, it is useful, like all vegetables, for improving intestinal transit and reducing the risk of colon cancer. It contains folic acid, which prevents neural tube defects during gestation, along with a good supply of other vitamins and minerals. An excellent food, in other words, that comes with one small adverse but entirely harmless effect. Within roughly 20 to 30 minutes of ingestion, the characteristic unpleasant odour released by its metabolites can already be detected in urine. Is this true for everyone? No.
It is estimated that around 40% of the population is able to perceive this characteristic smell, compared to a majority of people who are considered anosmic to that particular odour. These data come from a small number of studies on the topic and carry some bias, but they give us a sense of the scale: a large proportion of the population is simply unable to detect certain specific smells. The situation is complicated further by another variable: the inability of some people to produce detectable metabolites in their urine at all. If you can smell that characteristic asparagus odour in your urine, you are part not only of a minority with a functioning olfactory receptor, but also of a subset of the population that produces appreciable quantities of these volatile molecules. Perception and production are two independent traits.
The asparagus case is not an isolated one. Anosmia for androstenone, a molecule present in truffle, has long been documented. Androstenone binds to the olfactory receptor OR7D4. Those who carry the functional variant of this receptor find the smell of truffle unpleasant, similar to sweat; those who carry the hypofunctional variant find it pleasant, or do not perceive it at all.
On the subject of volatile metabolites: there is still no consensus in the scientific community as to which compound is directly responsible for the pungent smell. The most credible hypothesis to date is that asparagusic acid is the likely precursor, and that its metabolism gives rise to sulphur-containing compounds responsible for the olfactory characteristics of the urine. Among these, Methanethiol has been proposed as the primary candidate, although the smell is most probably produced not by a single molecule but by a narrow mixture of compounds.
Olfactory receptors form the largest gene family in the human genome, with approximately 400 members, each capable of binding specific odorous molecules. They belong to the large class of G protein-coupled receptors (GPCRs): molecules that translate an extracellular chemical signal into a response inside the cell. Each sensory neuron exposes on its surface only one of the nearly 400 olfactory receptors, and that receptor is tuned to a specific frequency of odours. This already gives us an indication of the degree of specificity and selectivity each receptor has towards particular odorous molecules.
From a genetic perspective, a genome-wide association study (a study that screens thousands of genomes to identify variants potentially associated with specific traits or diseases) identified as many as 871 distinct polymorphisms (frequent mutations in the population) with significant association values, all localised on chromosome 1 and the majority falling within the olfactory receptor 2 gene family. Minor mutations within these genes are sufficient to impair the ability of the resulting receptor to bind its ligand, that is, the odorous molecule.
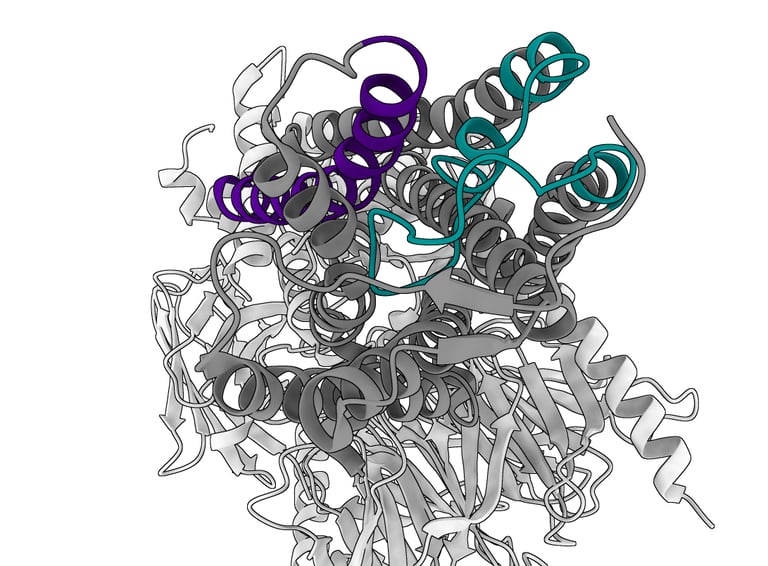
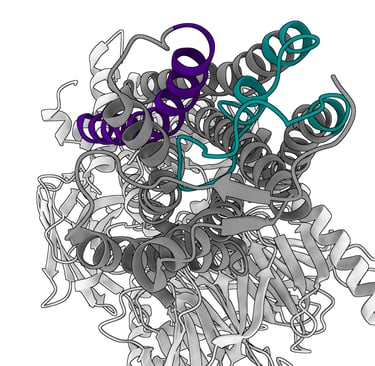
From a structural standpoint, olfactory receptors have long remained obscure proteins. As membrane proteins, they have limited affinity for water and require considerable laboratory effort to produce in quantities sufficient for the highly complex structural biology techniques needed to visualise them. It was only in 2023 that the first atomic-resolution structure of an olfactory receptor, OR51E2, was obtained. We can now use this structure as a basis for hypothesising how the olfactory receptor involved in perceiving the asparagus odour in urine, or any other olfactory receptor, might function, aided also by molecular modelling software.
In essence, the odorant fits into a cavity in the receptor sized to accommodate only that class of molecules. This binding triggers a conformational change in the receptor, which sets off a cascade of events whose ultimate outcome is the conscious perception of that smell.
Starting from the structural data of OR51E2, it is reasonable to speculate that the receptor involved in perceiving the pungent smell of urine after asparagus ingestion functions in a similar way. While the basic architecture and G protein coupling are probably conserved, the precise manner in which the binding pocket closes around the odorant, and the extent of the conformational movements that occur within the protein, could vary significantly across different olfactory receptor families precisely because they must recognise and accommodate molecules that can be very different from one another.
Contrary to what one might expect, olfactory receptors are not exclusive to the olfactory epithelium. OR51E2 itself has been found, for example, on the surface of prostate cancer cells, and is currently being studied as a potential therapeutic target because its activation inhibits cell proliferation. Olfactory receptors have also been identified in other tissues, such as the kidney, where they clearly do not serve an olfactory function. Olfr78 is capable of binding certain short-chain fatty acids produced by the metabolism of the gut microbiota. Its role is to mediate the secretion of renin, which in turn regulates blood pressure. A mechanism that reminds us how actively the gut microbiota participates in the physiology of the entire organism.
Let us return to asparagus. Behind the apparently trivial phenomenon of the olfactory perception of its urinary metabolites lies a more significant mechanism: the functioning of olfactory receptors in the broader sense. Understanding how olfactory receptors work inside and outside the nose may open new avenues in pharmacology. Whether or not you smell that odour after an asparagus meal tells you nothing of importance about your health. The biological mechanism that determines it, however, could tell us something very concrete about how cells perceive the chemical world around them.
FURTHER READING
Olfactory receptors belong to the large class of G protein-coupled receptors (GPCRs), which share a common general structure.
Picture a single chain of amino acids that crosses the cell membrane seven times, much like a needle and thread passing through a piece of fabric seven times. The portions that remain outside the membrane serve, on the extracellular side, to recognise the specific ligand, and on the intracellular side to relay the signal of successful binding to the interior of the cell. OR51E2 in particular possesses 7 α-helices that span the neuronal membrane, connected by loops, which are portions of the protein without a rigid structure.
In the specific case of OR51E2, ligand recognition depends on an arginine residue (R265), a highly conserved amino acid among members of this receptor family, located on the extracellular-facing portion of helix 6. This amino acid, which carries a positive charge, forms electrostatic interactions with the negatively charged carboxyl group of odorant molecules such as octanoate or propionate. In practice, this arginine anchors the odorant molecule as it inserts itself into the bundle of transmembrane helices (particularly between TM3 and TM6), which together form a binding pocket. Extracellular loop 2 (ECL2) acts as a molecular lid, partially covering this pocket and further stabilising the binding of the odorant’s carboxyl group through additional positively charged residues.
Odorant binding triggers a dramatic conformational change in the receptor: the extracellular portion of helix TM6 swings inward, closing the pocket over the odorant, while the intracellular portion moves outward to allow coupling with the G protein on the cytoplasmic side and the subsequent downstream signalling cascade. The signals triggered by odorant binding ultimately aim at depolarising the membrane of the olfactory neuron, generating a nerve impulse that is transmitted to the brain and enables the conscious perception of a specific smell.
References
Pelchat ML, Bykowski C, Duke FF, Reed DR. Excretion and perception of a characteristic odor in urine after asparagus ingestion: a psychophysical and genetic study. Chem Senses. 2011 Jan;36(1):9-17.
Yu Y, Ma Z, Pacalon J, Xu L, Li W, Belloir C, Topin J, Briand L, Golebiowski J, Cong X. Extracellular loop 2 of G protein-coupled olfactory receptors is critical for odorant recognition. J Biol Chem. 2022 Oct;298(10):102331.
Markt SC, Nuttall E, Turman C, Sinnott J, Rimm EB, Ecsedy E, Unger RH, Fall K, Finn S, Jensen MK, Rider JR, Kraft P, Mucci LA. Sniffing out significant “Pee values”: genome wide association study of asparagus anosmia. BMJ. 2016 Dec 13;355:i6071.
Choi C, Bae J, Kim S, Lee S, Kang H, Kim J, Bang I, Kim K, Huh WK, Seok C, Park H, Im W, Choi HJ. Understanding the molecular mechanisms of odorant binding and activation of the human OR52 family. Nat Commun. 2023 Dec 7;14(1):8060.
Pluznik JL, Protzko RJ, Gevorgyan H, Peterlin Z, Sipos A, Han J, Brunet I, Wan LX, Rey F, Wang T, Firestein SJ, Yanagisawa M, Gordon JI, Eichmann A, Peti-Peterdi J, Caplan MJ. Olfactory receptor responding to gut microbiota-derived signals plays a role in renin secretion and blood pressure regulation. Proc Natl Acad Sci U S A. 2013 Mar 12;110(11):4410-5.
Image
Cryo-EM structure of the olphactory receptor O51E2. Molecule visualized with ChimeraX. TM6 is highligthed in purple, EC2 loop in green.
